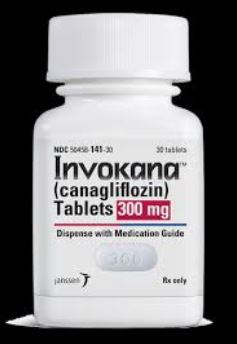
The U.S. Food & Drug Administration (FDA) approved Invokana as the first SGLT2 inhibitor that is used for the treatment of Type 2 diabetes in adults. Despite the drug being popular in a short span of time, it has some very serious side effects. The drug is used to reduce the blood glucose levels in the patients by preventing the absorption of sugar by the kidneys and then flush out through urine. It is the trade name of the drug canagliflozin and is an oral diabetic medicine.

The FDA has released warning about the harmful effects of the drug and this otherwise popular medicine had a large number of negative reports from the patients.
Canagliflozin, sold under the drug name Invokana, was introduced in the market for Type 2 diabetics and was meant to lower the blood sugar. It should never be used for Type 1 diabetes treatment. The FDA got hundreds of adverse reports about the invokana side effects and the victims claimed for invokana settlements.
The main complications cited in the allegations
The patients who filed for the lawsuits said that the following health risks are the main concern for those who take Invokana. Let’s have a look at the:
- Diabetic ketoacidosis is a dangerous one which is associated with Invokana. Over 14% of all diabetics have been hospitalized because of ketoacidosis and if it is not diagnosed or treated timely, it can lead to coma and death. According to the FDA, more than 20 cases of ketoacidosis were reported as early as within 15 months of Invokana being released in the market.
- Invokana is also linked with kidney damage. As the organs are responsible for filtering the waste products from the body, any malfunction to these organs can increase wastes in the blood stream and further lead to dialysis. The FDA has received more than a hundred reports of acute kidney injury of which 96 of the needed emergency hospitalization. Of them, more than half of the victims had acute kidney injury within one month of being exposed to the drug. With discontinuation of the medication, some patients saw better physical health but the FDA warns that renal injury is not reversible in all the conditions.
- One of the most hazardous health conditions of the invokana side effects is foot or leg amputation. In 2016, the FDA released a safety warning how the risk of amputation is far more in those patients taking invokana than those who take any other substitute for the drug. Peripheral arterial disease, nerve damage and poor blood circulation are the main reasons that cause this condition.
The milder side effects of the drug that are not reported in the invokana settlements include but not limited to:
- increased blood pressure
- frequent urge to urinate
- loss of appetite
- nausea
- bladder pain
- weight gain
In the court filings, the attorneys dispute that:
- Invokana is an unreasonably dangerous drug
- Johnson & Johnson was negligent in the manufacturing of the drug risking severe health risks
- The company failed to do proper clinical study and test of the medication
- They did not inform either the physicians or the patients about the different potential side effects of the drug
- Johnson & Johnson masked the evidence of the harmful side effects and misbranded the safety and effectiveness of the medication